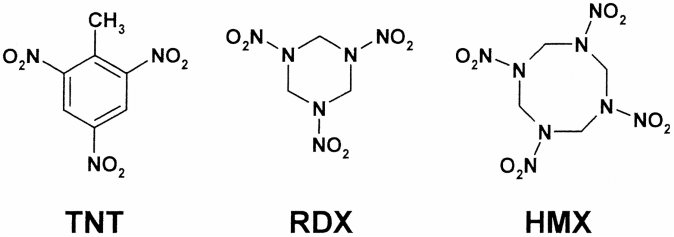
A Quick Summary of Trinitrotoluene
TNT is one of the most commonly used explosives for military and industrial applications. It is valued partly because of its insensitivity to shock and friction, which reduces the risk of accidental detonation, compared to other more sensitive high explosives such as nitroglycerin. TNT melts at 80 °C (176 °F), far below the temperature at which it will spontaneously detonate, allowing it to be poured as well as safely combined with other explosives. TNT neither absorbs nor dissolves in water, which allows it to be used effectively in wet environments. Additionally, it is stable compared to other high explosives. In order to initiate an explosion, TNT must first be detonated using a pressure wave from a more sensitive explosive called an explosive booster. Although blocks of TNT are available in various sizes (e.g. 250 g, 500 g, 1,000 g), it is more commonly encountered in synergistic explosive blends comprising a variable percentage of TNT plus other ingredients. Examples of explosive blends containing TNT include:
- Amatex: (ammonium nitrate and RDX)
- Amatol:(ammonium nitrate)
- Ammonal: (ammonium nitrate and aluminum powder plus sometimes charcoal).
- Baratol: ( barium nitrate and wax)
- Composition B ( RDX and paraffin wax)
- Composition H6
- Cyclotol (RDX)
- Ednatol
- Hexanite (hexanitrodiphenylamine).
- Minol
- Octol
- Pentolite
- Picratol
- Tetrytol
- Torpex
- Tritonal
The Uses Of TNT (trinitrotoluene)
TNT was first prepared in 1863 by German chemist Julius Wilbrand and originally used as a yellow dye. TNT can be safely poured when liquid into shell cases, and is so insensitive that in 1910, it was exempted from the UK's Explosives Act 1875 and was not considered an explosive for the purposes of manufacture and storage. TNT-filled armor-piercing shells would explode after they had penetrated the armor of British capital ships, whereas the British lyddite-filled shells tended to explode upon striking armor, thus expending much of their energy outside the ship. TNT was also used to fill filling naval mines, bombs, depth charges, and torpedo warheads with buster charges of crude grade B TNT with the color of brown sugar and requiring an explosive booster charge of granular crystallized grade A TNT for detonation.
What is The Chemical Formula For Trinitrotoluene
The chemical formula for trinitrotoluene is C6H2(NO2)3CH3. Upon Combustion trinitrotoluene detonation formula is
2 C7H5N3O6 → 3 N2 + 5 H2O + 7 CO + 7 C and 2 C7H5N3O6 → 3 N2 + 5 H2 + 12 CO + 2 C
2 C7H5N3O6 → 3 N2 + 5 H2O + 7 CO + 7 C and 2 C7H5N3O6 → 3 N2 + 5 H2 + 12 CO + 2 C
TNT's Explosive Character
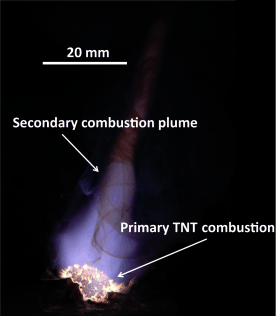
The reaction is exothermic but has a high activation energy. Because of the production of carbon, TNT explosions have a sooty appearance. Because TNT has an excess of carbon, explosive mixtures with oxygen-rich compounds can yield more energy per kilogram than TNT alone. During the 20th century, amatol, a mixture of TNT with ammonium nitrate was a widely used military explosive. Detonation of TNT can be done using a high velocity initiator or by efficient concussion. But for many years, TNT used to be the reference point for the Figure of Insensitivity. TNT had a rating of exactly 100 on the F of I scale. The reference has since been changed to a more sensitive explosive called RDX, which has an F of I of 80.
Energy Content
Pentolite is a high explosive used for military and civilian purposes e.g. warheads and booster charges. TNT is reported to contain 2.8 mega joules per kilogram explosive energy. The actual heat of combustion is 14.5 megajoules per kilogram, which requires that some of the carbon in TNT react with atmospheric oxygen, which does not occur in the initial event.The explosive energy utilized by NIST is 4184 J/g (4.184 MJ/kg). The energy density of TNT is used as a reference-point for many other types of explosives, including nuclear weapons, the energy content of which is measured in kilotons (~4.184 Tera-joules) or megatons (~4.184 Peta joules) of TNT equivalent. For comparison, gunpowder contains 3 megajoules per kilogram, dynamite contains 7.5 megajoules per kilogram, and gasoline contains 47.2 megajoules per kilogram (though gasoline requires an oxidant, so an optimized gasoline and O2 mixture contains 10.4 megajoules per kilogram).
Safety And Toxicity
TNT is poisonous, and skin contact can cause skin irritation, causing the skin to turn a bright yellow-orange color. People exposed to TNT over a prolonged period will tend to experience anemia and abnormal liver functions/activity. Blood and liver effects, spleen enlargement and other harmful effects on the immune system have also been found in animals that ingested or breathed trinitrotoluene. There is evidence that TNT adversely affects male fertility. TNT is listed as a possible human carcinogen, with carcinogenic effects demonstrated in animal experiments (rat), although effects upon humans so far amount to none [according to IRIS of March 15, 2000]. Consumption of TNT produces red urine through the presence of breakdown products and not blood as sometimes believed.
Some military testing grounds are contaminated with TNT. Waste water from munitions programs including contamination of surface and subsurface waters may be colored pink because of the presence of TNT. Such contamination, called "pink water", may be difficult and expensive to remedy.
TNT is prone to exudation of dinitrotoluenes and other isomers of trinitrotoluene. Even small quantities of such impurities can cause such effect. The effect shows especially in projectiles containing TNT and stored at higher temperatures, e.g. during summer. Exudation of impurities leads to formation of pores and cracks (which in turn cause increased shock sensitivity). Migration of the exudate liquid into the fuze screw thread can form fire channels, increasing the risk of accidental detonations; fuze malfunction can result from the liquids migrating into its mechanism. Calcium silicate is mixed with TNT to mitigate the tendency towards exudation.
Some military testing grounds are contaminated with TNT. Waste water from munitions programs including contamination of surface and subsurface waters may be colored pink because of the presence of TNT. Such contamination, called "pink water", may be difficult and expensive to remedy.
TNT is prone to exudation of dinitrotoluenes and other isomers of trinitrotoluene. Even small quantities of such impurities can cause such effect. The effect shows especially in projectiles containing TNT and stored at higher temperatures, e.g. during summer. Exudation of impurities leads to formation of pores and cracks (which in turn cause increased shock sensitivity). Migration of the exudate liquid into the fuze screw thread can form fire channels, increasing the risk of accidental detonations; fuze malfunction can result from the liquids migrating into its mechanism. Calcium silicate is mixed with TNT to mitigate the tendency towards exudation.
Bio-Degradation/Disposal Issues
The ligninolytic physiological phase and manganese peroxidase system of fungi can cause a very limited amount of mineralization of TNT in a liquid culture; though not in soil. An organism capable of the remediation of large amounts of TNT in soil has yet to be discovered. Both wild and transgenic plants can premeditated explosives from soil and water.
Physical and Chemical Properties
Physical Properties:
Color: Yellow
Physical State: Mono-clinic needles
Molecular Weight: 227.13
Chemical Properties:
Explosive temperature: 464 F
Vapor pressure at 20 C: 1.99*10^-4 mm-Hg
Flash point: Explodes
Color: Yellow
Physical State: Mono-clinic needles
Molecular Weight: 227.13
Chemical Properties:
Explosive temperature: 464 F
Vapor pressure at 20 C: 1.99*10^-4 mm-Hg
Flash point: Explodes